Regenerative Calcium Management
Calcium is often called the “trucker of all minerals” and is essential for building strong cell walls, supporting root development, and improving crop quality. But in regenerative agriculture, its role goes far beyond plant nutrition. Calcium is a important for soil structure, water movement, nutrient balance, and biological health.
Managing calcium regeneratively means looking at the whole system, from the soil reserves and cation balance, to plant uptake, transport, and cycling through biology. It’s not just about adding more calcium; it’s about ensuring it’s in the right form, at the right balance, and supported by the soil biology that makes it continuously available.
This guide brings together everything you need to know about calcium in a regenerative system, including:
The role of calcium in plants and why it’s critical at every growth stage.
How calcium behaves in the soil and the factors affecting its availability.
Interpreting soil and sap tests to assess calcium supply and uptake.
Fertiliser options and how to choose the right source for your needs.
Balancing application rates to optimise soil structure and nutrient efficiency.
Advanced regenerative strategies
Whether you’re dealing with poor soil structure, plant quality issues, or nutrient imbalances, this comprehensive approach will help you manage calcium in a way that strengthens your soils, feeds your crops, and reduces long-term input costs.
First watch this video below from our YouTube Channel (And subscribe).
The Role of Calcium in Plants
Calcium is often called the “trucker of all minerals” in plant nutrition. It’s not just a nutrient; it’s a driver of plant quality, resilience, and growth. From building strong cell walls to boosting pest and disease resistance, calcium is fundamental to plant health and productivity.
1. Strengthening Cell Walls and Membranes
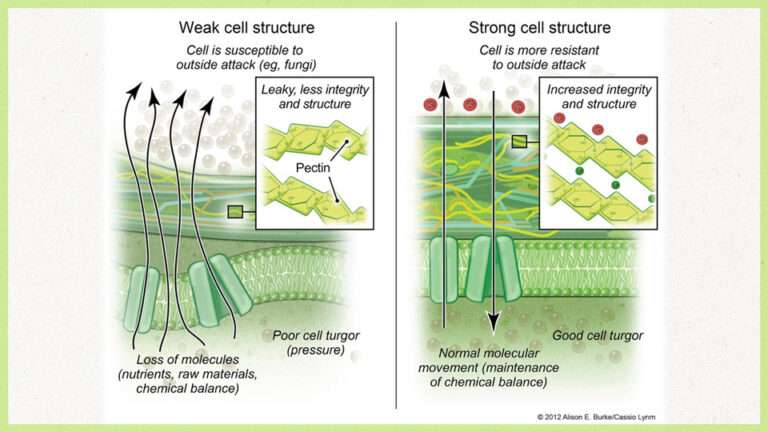
Calcium is a key component in developing strong, functional cell walls and membranes. Inside plant cells, compounds called pectins form part of the wall’s structure. Without calcium, these pectins remain loose and disorganised, leading to weak cell walls. Calcium’s double positive charge allows it to bind two pectin molecules together, creating a reinforced structure, much like steel rebar strengthens concrete.
Strong cell walls have two big benefits:
Reduced water loss – Firm walls limit leakage, helping regulate transpiration.
Improved nutrient transport – A strong “infrastructure” ensures minerals move efficiently throughout the plant.
This structural strength is essential for post-harvest quality. Issues like blossom end rot in tomatoes are often more about calcium deficiency than pests or diseases. When cell walls are weak, microbes can invade easily, reducing storage life and market quality.
2. Building Pest and Disease Resistance
A rigid cell wall acts as the plant’s first line of defence against fungal pathogens such as moulds, rots, blights, and mildews. Calcium-reinforced tissues are harder for pests and diseases to penetrate, giving crops a natural resilience that reduces reliance on chemical controls.
3. Driving Cell Division and Elongation
Calcium isn’t only about structure, it’s vital for growth. New cells require calcium to build their walls, meaning it directly influences cell division and elongation. Unlike nitrogen, which can sometimes create rapid but low-quality growth, calcium supports strong, high-quality tissue formation.
The benefits are especially clear in roots. Research shows that adequate calcium supply can increase root extension length fourfold compared to deficient plants. Longer, more extensive roots mean greater access to water and nutrients, and improved interaction with beneficial microbes.
4. Supporting Reproductive Development
Critical growth stages such as bud formation, flowering, and early fruit set rely on calcium-driven cell division. Ensuring adequate calcium during these periods can maximise fruit size, improve firmness, and enhance overall crop yield potential. These are the critical points of influence that we target to optimise yields.
Calcium in the Soil
Calcium is one of the most abundant elements in many soils, yet plants can still suffer from calcium deficiency. That’s because the amount present is not the same as the amount available. Understanding how calcium exists in the soil and how it interacts with other minerals, pH, and soil structure, is essential for managing it effectively.
1. The Three Forms of Calcium in Soil
Calcium occurs in three main forms, each playing a different role:
Mineral Calcium – Locked in soil minerals such as calcium feldspars, often in the parent rock material. This is not directly available to plants but can be released slowly through weathering or biological activity. Basalt-derived soils, for example, tend to have higher calcium levels than sandy, low-mineral soils.
Exchangeable Calcium – Attached to the negatively charged sites on clay particles or organic matter. This is a key plant-available form, as roots can swap hydrogen ions for calcium and absorb it directly.
Soluble Calcium – Dissolved in the soil water (soil solution) and immediately available for plant uptake. This pool is in dynamic equilibrium with exchangeable calcium.
2. Interactions with Other Cations
Calcium competes with other positively charged minerals (cations) such as magnesium, potassium, sodium, ammonium, iron, and manganese for uptake and exchange sites. Excessive magnesium or ammonium, for example, can strongly suppress calcium availability. This is why soil test interpretation should always consider the balance between cations, not just total amounts.
3. The Role in Soil Structure and Flocculation
Beyond plant nutrition, calcium is a important for soil structure. On clay particles, calcium encourages flocculation, the clumping together of tiny clay particles into stable aggregates. This improves water infiltration, aeration, and root penetration.
Magnesium also binds clays, but its stronger charge attracts more water, pushing particles apart and reducing aggregation. Sodium is even worse, causing dispersion, where clay particles separate and clog pores, leading to crusting and poor infiltration. Maintaining adequate calcium relative to magnesium and sodium is therefore important for healthy soil structure.
4. Calcium and pH
Calcium is often supplied through liming materials such as calcium carbonate (lime), which raise soil pH by neutralising excess hydrogen ions. This not only reduces acidity but also improves the availability of other nutrients. However, not all calcium sources affect pH — gypsum, for example, supplies calcium and sulfur without changing soil acidity, making it valuable for sodic or alkaline soils.
5. Holding Capacity and Soil Type
Clay and organic matter provide the “holding sites” for calcium. Sandy soils, with fewer holding sites, are more prone to calcium leaching and deficiency. Increasing soil organic matter can help these soils retain calcium longer, reducing the risk of seasonal shortages.
6. Cycling Calcium from the Subsoil
In many soils, especially duplex types common in Australia, calcium can accumulate in the subsoil over time. Deep-rooted plants including certain cover crops and even some weeds can mine this subsoil calcium and return it to the surface via plant residues, creating a natural nutrient recycling loop.
Calcium Levels in Soil Tests
Soil tests are the foundation of good calcium management. Understanding these results is essential for making cost-effective and targeted applications.
1. Key Calcium Measurements in Soil Tests
The comprehensive soil tests we use report calcium in three forms:
Total Mineral Calcium – The full amount locked up in soil minerals. This isn’t all plant-available, but it’s an important reserve. Basalt and other mafic parent materials tend to show higher levels than light sandy soils.
Exchangeable Calcium – The calcium held on the cation exchange sites of clay particles and organic matter. This is a major source of plant-available calcium.
Soluble Calcium – Calcium dissolved in the soil water and immediately available for uptake. This form is constantly moving between the soil solution and exchangeable pool.
2. Ideal Ranges for Calcium
Ideal calcium levels depend on soil texture (clay, loam, or sand) and organic matter. As a guide:
Mineral calcium – At least ~1,000 ppm on sandy soils, with higher levels desirable for clays, up to 10,000ppm.
Exchangeable calcium – ~1,000 ppm on sands, up to 3,000+ ppm on heavier clays.
Soluble calcium – Around 200ppm for sandy soils and up past 1000ppm for clay soils.
These are baseline indicators. In heavier soils, more calcium is needed because there are more negatively charged sites to fill. In sandy soils, lower targets are used, but leaching risk is higher.
In addition to these tests and tests, we often use Haney Soil Tests, which aims to have calcium at 500ppm
3. % Calcium – The Percentage of Sites Occupied by Calcium
Many soil tests also express calcium as a percentage of effective cation exchange capacity (eCEC). This figure helps assess balance with other cations like magnesium, potassium, and sodium.
General targets:
60–80% calcium for most soils
Slightly lower in sands (which are already well-aerated)
Higher in clays (to promote good flocculation and aggregation)
Falling well below these ranges may indicate a deficiency or poor soil structure, while excessive levels can antagonise other nutrients.
4. The Calcium–Magnesium Ratio Debate
The “Ca:Mg ratio” is often discussed in soil balancing circles. Research confirms calcium improves soil structure by flocculating clay, but the ratio’s direct effect on yield is less certain. Farmers often see quality and structural benefits when improving the ratio, especially where magnesium levels are high and soils are tight or poorly draining.
In our consulting, we consider the Ca:Mg but tend not to give too much priorty to the ratio, unless it is significantly out of balance.
Typially the Ca:Mg will range from 3:1 in sands to 6:1 in clays.
Calcium Fertiliser Options
Supplying calcium to your crops isn’t just about adding more of the mineral, it’s about choosing the right source for your soil type, pH, crop needs, and overall nutrient balance. Different fertilisers deliver calcium in different ways, and each comes with its own benefits, limitations, and ideal use cases.
1. Soil Amendments – Building Long-Term Calcium Supply
These products are generally applied to the soil to build or restore calcium reserves.
Lime (Calcium Carbonate)
Calcium content: ~40%
Best for: Acidic soils needing pH correction
Benefits: Neutralises acidity, increases calcium availability, improves nutrient uptake
Considerations: Apply at least 1 tonne/ha for economic viability; avoid exceeding ~4 tonnes/ha in a single application to prevent antagonising other minerals.
Dolomite (Calcium Magnesium Carbonate)
Calcium content: ~22%, plus magnesium
Best for: Acidic soils low in both calcium and magnesium
Benefits: Corrects pH while balancing the Ca:Mg ratio in sandy soils
Considerations: Avoid if magnesium levels are already high.
Gypsum (Calcium Sulfate)
Calcium content: ~23% plus sulfur
Best for: Sodic soils, soils with high magnesium, or alkaline soils where pH change isn’t desired
Benefits: Improves soil structure via flocculation, displaces sodium, supplies sulfur
Considerations: More soluble than lime, so apply carefully to avoid excessive cation stripping; generally limit to <800 kg/ha per application.
Soft Rock Phosphate
Calcium content: ~19% (varies), plus phosphorus and silica
Best for: Soils deficient in both phosphorus and calcium
Benefits: Supplies multiple minerals and has a mild liming effect (1/5th of lime)
Considerations: Works best in biologically active soils to release nutrients.
Guano
Calcium content: ~30%, plus phosphorus and trace elements
Best for: Starter fertiliser at planting
Benefits: More immediately available than lime or dolomite; supports early root and cell wall development
Considerations: Higher cost means it’s best used in targeted applications.
2. Foliar and Fast-Acting Calcium Sources
These products are ideal for addressing short-term deficiencies or supplying calcium at critical growth stages.
Calcium Acetate
Best for: Foliar sprays during high-demand periods
Benefits: Contains carbon, aiding plant energy metabolism; highly available
Considerations: Can be combined with fulvic acid for improved uptake.
Calcium Nitrate
Best for: Crops needing both nitrogen and calcium
Benefits: Highly soluble, rapid response
Considerations: Nitrate form can cause dilution growth; best used with strategies to convert nitrate into amino acids.
Calcium Chloride
- Not use since this can easily cause leaf burn and excess chlorides will negatively affect the plant.
Micronised Lime, Gypsum, or Guano
Best for: Foliar feeding where the benefits of the other sources are desired.
Considerations: Requires excellent spray tank agitation to prevent settling.
In regenerative systems, the goal is to build a stable calcium reserve in the soil while using targeted in-season applications to address critical growth stages or prevent quality losses. This means combining long-term amendments with foliar tools without becoming dependent on constant foliar feeding.
Calcium in Plant Sap
Calcium is one of the most important minerals for plant structure, quality, and resilience but understanding its status inside the plant isn’t as simple as looking at one number. Sap testing gives real-time insight into calcium uptake and movement, helping you fine-tune nutrition before problems show in the paddock.
1. Why Sap Testing is Valuable for Calcium
Soil tests tell you if calcium is present in the soil, but they don’t confirm if the plant is actually taking it up. Sap testing, especially differential sap testing (comparing young and old leaves), shows how calcium is moving inside the plant and whether supply is keeping up with demand.
A typical approach is to test:
Old leaves – where immobile minerals like calcium accumulate
Young leaves – which rely on a fresh supply of calcium from the roots
If old leaves have high calcium but young leaves are low, it’s a sign the plant is struggling to draw in enough new calcium from the soil.
2. Calcium Mobility
Calcium moves only through the xylem with the transpiration stream, and once deposited in tissues, it’s locked in place. Unlike mobile nutrients such as nitrogen or potassium, calcium cannot be relocated from older tissues to new growth.
This means:
A steady supply throughout the season is critical
Any disruption in uptake (dry soils, root damage, antagonism from other nutrients) will quickly show up in new leaves, buds, flowers, or fruit
Foliar sprays can provide a short-term boost, but they don’t fix the underlying soil uptake problem
3. Nutrient Interactions to Watch
Several nutrients can antagonise calcium uptake if they’re in excess:
Magnesium (Mg) – Competes with calcium for exchange sites and uptake; high Mg soils often have tight structure and poor root penetration.
Potassium (K) – Excessive K, particularly from high fertiliser rates, can reduce calcium absorption.
Sodium (Na) – Displaces calcium from exchange sites and can degrade soil structure.
Ammonium (NH₄⁺) – One of the strongest competitors for calcium uptake; heavy ammonium fertiliser use can sharply reduce calcium availability.
Iron (Fe) and Manganese (Mn) – High levels can also restrict calcium absorption.
On the flip side, boron plays a supporting role, it’s essential for calcium transport within the plant. Low boron can cause a calcium deficiency even if soil calcium is adequate.
4. Reading Sap Test Results for Calcium
When assessing sap tests:
Look at the young-to-old leaf ratio – Large gaps (e.g. 1,000 ppm in old leaves vs. 700 ppm in young) indicate an uptake problem.
Compare to soil test data – Low soil reserves point to a supply issue; adequate soil levels with poor plant uptake suggest root or soil biology constraints.
Consider crop stage – Critical points like cell division, early fruit set, and rapid growth phases require peak calcium supply.
Calcium Balancing and Application Rates
Supplying calcium isn’t just about topping up a deficiency, it’s often about balancing calcium with other cations in the soil to improve plant health, soil structure, and nutrient uptake. The right application rate depends on soil test data, texture, and your target balance.
1. Why Calcium Balancing Matters
Calcium competes with other positively charged nutrients (cations) like magnesium, potassium, sodium, and ammonium for exchange sites in the soil. If calcium is too low, soil structure suffers and plant uptake drops. If it’s too high, other nutrients can become unavailable.
Balancing calcium improves:
Soil flocculation – clay particles aggregate, improving aeration and infiltration
Nutrient uptake – especially potassium and micronutrients
Plant quality – stronger cell walls, improved disease resistance
2. Sufficiency Levels
Similar to other methods, calculate the difference between ideal levels and current levels to determine how much to apply. This method in combination with the balancing method is important.
3. The Balancing Process
To calculate a balancing application rate, you’ll need:
Effective Cation Exchange Capacity (ECEC) – from your soil test, including hydrogen and aluminium
Current calcium base saturation (%) – the percentage of cation exchange sites occupied by calcium
Ideal calcium base saturation (%) – typically 60–80% depending on soil texture (lower for sands, higher for clays)
Example calculation:
Soil ECEC = 14 cmol(+)/kg
Current Ca base saturation = 50%
Target = 75%
Difference = 25%
Convert the percentage to parts per million (ppm):
14×0.25×200=700ppm
Convert ppm to kilograms per hectare (at 10 cm depth, bulk density 1.4):
700×1.4=980 kg Ca/ha required
Convert into product
- 980/product ca% = product to apply in kg/ha
If using lime (~40% calcium)
980÷0.40=2,450 kg lime/ha
3. Choosing the Right Product for Balancing
Lime (CaCO₃) – Best for acidic soils; raises pH and adds calcium
Dolomite (CaMg(CO₃)₂) – Adds both calcium and magnesium; use only if Mg is also low
Gypsum (CaSO₄·2H₂O) – Best for sodic or high-Mg soils; doesn’t affect pH
Soft rock phosphate or guano – Can be used if phosphorus is also needed
4. Practical Tips for Application Rates
Split large applications – If calculated rates exceed ~4 t/ha of lime or 800 kg/ha of gypsum, split over 3–5 years to avoid antagonising other nutrients
Match to soil type – Sandy soils can’t hold as much calcium; use smaller, more frequent applications to reduce leaching
Don’t chase perfection – Close to the ideal range is good enough; exact targets aren’t necessary for productive crops
Consider pH effects – Lime and dolomite raise pH; gypsum doesn’t. Typically, we use this method to determine liming rates to raise pH since this factors the ability of the soil to release H into the soil solution, however, it is likely that this underestimates liming applications. This may mean that lime will need to be applied but this prevents overapplying lime.
5. Balancing vs. Maintenance Applications
Once the calcium base saturation is in range, switch to maintenance applications to replace what’s lost through crop removal, leaching, or soil exchange. In many systems, this means smaller, regular applications rather than large, infrequent ones.
How to Manage Calcium Regeneratively
In regenerative agriculture, calcium management isn’t just about correcting deficiencies, it’s about creating a balanced, biologically supported supply that feeds plants continuously, strengthens soils, and supports nutrient cycling. This approach builds resilience, reduces dependency on constant inputs, and supports long-term soil health.
1. Focus on Soil Health First
Calcium uptake depends on more than its concentration in the soil, it relies on the condition of the root zone:
Structure: Aggregated soils with stable pores allow roots and water to reach exchangeable calcium.
Biology: Beneficial microbes and fungi help dissolve mineral-bound calcium and deliver it directly to plants.
Moisture: Adequate soil moisture is essential for calcium movement in the xylem.
Without these conditions, even calcium-rich soils can produce crops with deficiency symptoms.
2. Maintain Cation Balance
Calcium competes with magnesium, potassium, sodium, ammonium, iron, and manganese for uptake. Excesses of these cations can block calcium movement into the plant. Regenerative management means:
Avoiding excessive potassium or ammonium fertiliser use.
Correcting excess magnesium or sodium through amendments like gypsum.
Using targeted applications to bring calcium into the ideal base saturation range (generally 60–80%, depending on soil type).
3. Support Calcium Transport with Boron
Boron plays a crucial role in the structural integrity of cell walls and in moving calcium within the plant. Without adequate boron, even good calcium supply won’t reach developing tissues effectively.
Maintain boron levels via soil and foliar applications as required.
Monitor with sap tests to avoid both deficiency and toxicity.
4. Build a Stable Reserve in the Soil
Long-term calcium supply comes from stable, slow-release sources:
Lime or dolomite for acidic soils, correcting both pH and calcium levels.
Gypsum for alkaline or sodic soils where pH change is not desired.
Soft rock phosphate or guano to supply calcium along with phosphorus and trace minerals.
These inputs act as a calcium “bank” that plants can access over time.
5. Harness Mycorrhizal Fungi for Calcium Uptake
Mycorrhizal fungi extend root access to otherwise unreachable soil calcium, particularly from mineral particles. In regenerative systems:
Avoid unnecessary fungicides that damage mycorrhizal populations.
Maintain living roots year-round through cover crops and pasture.
Reintroduce mycorrhizal inoculants if the system has been degraded.
6. Use Calcite-Solubilising Bacteria
Certain bacterial species can dissolve calcium carbonate (calcite) in the soil, releasing calcium into plant-available forms. These microbes also help buffer pH and unlock other nutrients. Strategies include:
Applying microbial inoculants with calcite-solubilising species.
Providing them with organic carbon sources to fuel their activity.
7. Increase Soil Carbon to Improve Calcium Holding
Building soil organic carbon enhances calcium management by:
Increasing the cation exchange capacity (CEC) and therefore the soil’s ability to hold calcium.
Feeding soil biology that helps solubilise and cycle calcium.
Improving soil structure for better root exploration and moisture retention.
8. Use the Rhizophagy Cycle
The rhizophagy cycle is a plant–microbe process where roots absorb microbes carrying nutrients including calcium into root cells. The plant then extracts the nutrients and releases the microbes back into the soil to collect more. This natural nutrient shuttle is supported by:
Maintaining diverse microbial communities.
Reducing synthetic inputs that suppress beneficial bacteria.
Keeping living roots in the soil year-round.
9. Cycle Calcium with Deep-Rooted Plants
Species like chicory, lucerne, and certain brassicas mine calcium from subsoil layers and return it to the surface in plant residues. Diverse cover crops increase rooting depths and enhance the biological cycling of calcium and other nutrients.
10. Apply Foliar Calcium Strategically
Foliar calcium in the form of acetate, nitrate, or micronised minerals is best used:
During high-demand stages like flowering, fruit set, and rapid cell division.
As a short-term quality protection measure, while addressing soil supply for the long term.
Building a Resilient Calcium Cycle
Calcium management in regenerative agriculture is not a one-off fix, it’s an ongoing process of balancing minerals, supporting soil biology, and aligning nutrient supply with plant demand. By understanding calcium’s role in both soil and plant systems, you can move beyond reactive applications and towards a self-sustaining calcium cycle.
The regenerative approach focuses on:
Maintaining the right calcium balance with other cations
Supporting biological partners like mycorrhizal fungi and calcite-solubilising bacteria
Using boron, soil carbon, and plant diversity to improve uptake and cycling
Applying foliar calcium only as a targeted, in-season tool
When calcium is managed this way, soils become more stable and better structured, roots explore deeper, plants grow stronger, and yields improve, without the constant churn of high-cost fertiliser inputs. Over time, your system shifts from dependency to resilience, with calcium continually recycled through biology, plants, and soil.
By applying these principles, you’re not just feeding a crop, you’re building a healthier, more productive farm ecosystem.







