Regenerative Nitrogen Management
Nitrogen (N) is one of the most critical and most mismanaged nutrients in agriculture. While conventional systems often rely heavily on synthetic nitrogen fertilisers, this approach can lead to nutrient losses, environmental damage, pest pressure, and declining soil health.
In regenerative farming, nitrogen management is approached differently. The goal isn’t just to apply less, it’s to manage nitrogen more efficiently, biologically, and in symbiosis with plant and soil systems. This complete guide brings together the key principles of regenerative nitrogen management, helping you make better decisions that support productivity, profitability, and long-term sustainability.
In this guide, we’ll explore:
The role of Nitrogen in plants
How nitrogen behaves in the soil
How plants metabolise nitrogen into proteins
Different types of nitrogen fertilisers and how to use them
How to build a nitrogen budget for your farm
- How to test nitrogen using both soil and sap analysis
Ways to increase nitrogen use efficiency
Alternative nitrogen sources beyond synthetic Nitrogen
How nitrogen affects pest and disease pressure
Whether you’re looking to reduce your inputs, improve plant health, or transition to a more regenerative system, this guide gives you the practical insights and tools to take control of your nitrogen strategy.
First, watch this video from our YouTube Channel Agresol (and subscribe!)
Role of Nitrogen in the Plant
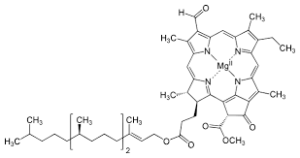
Nitrogen plays a vital role in plant growth and productivity, acting as a key building block of proteins, enzymes, and chlorophyll. It is essential for photosynthesis, cell division, and the development of new tissue, particularly in rapidly growing parts such as shoots and leaves. Nitrogen also influences the plant’s ability to produce amino acids, which are the foundation of proteins, and directly affects yield and biomass accumulation. A deficiency in nitrogen often results in stunted growth, pale or yellowing leaves (chlorosis), and reduced productivity, while excessive nitrogen can lead to excessive vegetative growth at the expense of reproductive development. In the image below is chlorophyll, it is the component in photosynthesis that captures light, you’ll notice that each chlorophyll contains 4 N at the ceneter.
Typically either nitrogen or water will be a limiting factor for yield and so it is very important it is manged effectively to prevent yield losses.
Nitrogen in the Soil
Nitrogen is a cornerstone nutrient in crop production. Yet, it’s also one of the most misunderstood and mismanaged, often leading to inefficiencies, environmental damage, and imbalanced plant nutrition. In regenerative systems, understanding how nitrogen behaves in the soil is the first step to managing it more effectively and sustainably.
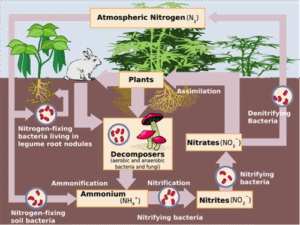
The image below is the classic nitrogen cycle, which for the most part is correct except that plants can absorb Ammonium AND organic N as well.
The Forms of Nitrogen in Soil
Nitrogen exists in two broad forms:
1. Inorganic Nitrogen
These are the plant-available forms most conventional systems rely on:
Ammonium (NH₄⁺) – A positively charged (cation) form that can bind to negatively charged soil particles such as clay and organic matter. This makes ammonium relatively stable and less prone to leaching. However, it can volatilise as ammonia gas if left on the soil surface, especially in alkaline conditions.
Nitrate (NO₃⁻) – A negatively charged (anion) form that does not bind to soil particles, making it highly mobile and prone to leaching or denitrification (conversion to gas). While readily plant-available, nitrate is also the most easily lost form of nitrogen.
2. Organic Nitrogen
This form is bound to carbon compounds and includes:
Amino acids
Amino sugars
Microbial biomass
Decomposing organic matter
Organic nitrogen is largely immobile, stable, and cannot leach like nitrate. It becomes available to plants through mineralisation, the microbial conversion of organic forms into ammonium and nitrate. Interestingly, recent research has confirmed that plants can directly absorb amino acids, making this a more efficient pathway for nitrogen uptake in biologically active soils.
Ideally we want the majority of nitrogen in the organic form to prevent nitrogen losses and improve nitrogen efficiency. Small amounts of nitrate and ammonium is important but shouldn’t be in excess.
Soil Testing For Nitrogen
Nitrogen management is only as good as the information behind it. Too often, fertiliser decisions are made based on guesswork, tradition, or fear of underperforming crops. But in regenerative systems, we use evidence-based testing to make confident, efficient nitrogen decisions.
Most nitrogen losses happen because fertiliser is applied without understanding actual plant needs or soil capacity. This leads to:
Wasted inputs
Leaching and volatilisation
Excess nitrate in sap (which attracts pests)
Poor conversion into proteins
With proper testing, you can:
Reduce nitrogen rates without compromising yield
Pinpoint the best form and timing of N
Improve efficiency and plant health
We suggest taking a soil test every season prior to planting (for annual crops) or the break of dormancy (for perennial crops) to inform nitrogen management.
Most soil tests record inorganic N (NO3- and NH4+), this is useful for determining nitrogen supply to the crop. However, as mentioned above, a very large pool of N exists in the organic component of the soil and rarely is included in soil test reports.
A haney soil test will provide measured water-extractable organic N as well as inorganic N. This is very important as it allows us to consider the complete nitrogen story in our soils. In our consulting work at Agresol, we use the haney soil test as our prefered soil testing method for nitrogen management.
On a soil test we like to see:
Nitrate (target ~13 ppm)
Ammonium (target ~18 ppm)
Water-extractable organic N (target 50+ ppm)
General Interpretation:
Low nitrate and ammonium = likely N deficiency
Low organic N = poor biological nitrogen fixation or nutrient cycling
High nitrate and ammonium = over-application and can lead to reduced supply of other minerals and likely pest and disease pressure.
- Application rate = (Ideal level (ppm) – Current supply (ppm)) x bulk density x depth (cm)/10
Nitrogen Metabolism in Plants
Nitrogen is essential for plant growth but simply applying nitrogen isn’t enough. For that nitrogen to truly benefit the crop, it must be efficiently converted into proteins through a tightly regulated sequence of metabolic steps. This is where nitrogen metabolism comes in.
Understanding how nitrogen is metabolised in the plant is critical for improving efficiency, reducing losses, and avoiding pest issues. In regenerative agriculture, we focus not just on how much nitrogen is applied, but how well the plant can use it and the required minerals to complete this process.
This complex process can be seen in the image below.
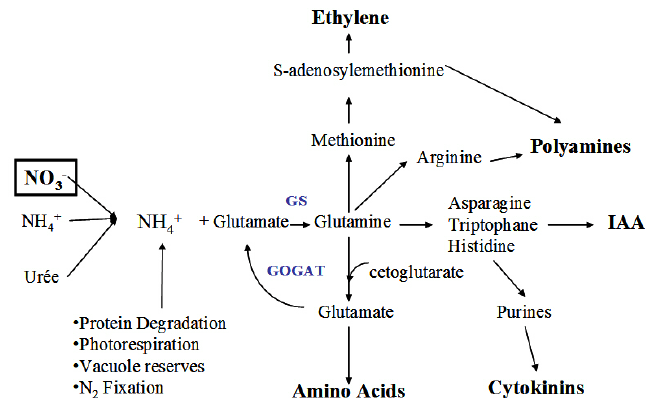
Step 1: How Plants Absorb Nitrogen
Plants can absorb nitrogen in several forms:
Nitrate (NO₃⁻) – A common fertiliser form, but requires conversion inside the plant into ammonium before being converted into amino acids.
Ammonium (NH₄⁺) – Can be absorbed and used more directly, but toxic in excess.
Urea – Urea can be directly absorbed by the plant either via the roots or the leaves.
Amino acids – The most efficient form, directly used by plants.
Regardless of the entry point, all nitrogen must eventually be transformed into amino acids, the building blocks of proteins, before the plant can use it for growth and function.
Nitrate and urea must be converted into ammonium before being converted into amino acids.
Typically nitrate will be transported to the laves to be converted into ammonium and then amino acids whereas ammonium will be converted into amino acids in the roots.
Step 2: Converting Into Ammonium
Nitrate must be converted into ammonium before it can enter amino acid metabolism:
Nitrate → Nitrite (via nitrate reductase)
Nitrite → Ammonium (via nitrite reductase)
This process uses a significant amount of plant energy—15 ATP per nitrate molecule. For context, a full cycle of photosynthesis produces around 18 ATP. Some estimates suggest that 24% of the root’s energy can go into converting nitrate, this can dramatically reducing yield potential if nitrate is the dominant N source.
These enzymes also rely on micronutrients:
Molybdenum and iron for nitrate reductase
Iron and sulphur for nitrite reductase
Deficiencies in these minerals stall the entire process, leaving nitrate sitting in plant sap, where it can attract pests and lead to imbalances.
Likewise, Urea must also be converted into ammonium. This requires the enzyme Urease which contains Nickel.
Step 3: Ammonium into Glutamate
Once in ammonium form (either absorbed directly or converted from nitrate/urea), the plant must convert ammonium into an amino acid called glutamate. This is the key nitrogen donor for most other amino acids.
This step requires:
Magnesium or manganese for the glutamine synthase enzyme
Sugars from photosynthesis to provide carbon skeletons and energy
Without enough photosynthates (sugars), ammonium builds up in the plant, leading to poor protein synthesis and increased pest pressure. This is why monitoring Brix levels is so critical in sap tests.
Step 4: Building Amino Acids and Proteins
Once glutamate is formed, the plant constructs all other amino acids, including:
Sulphur-based amino acids (like methionine and cysteine), requiring sulphur
Other essential amino acids, using various enzymes and cofactors
Amino acids then link together into proteins via polypeptide chains a process that requires:
Phosphorus
Magnesium
Zinc
Boron
Potassium
Manganese
This is where nitrogen is fully functional, used in enzymes, plant structures, and metabolic processes.
Why Organic Nitrogen Is More Efficient
When plants absorb amino acids directly (via organic nitrogen pathways or microbial exudates), they bypass all the conversion steps, saving up to 20+ ATP per molecule and preserving photosynthetic energy for yield and resilience.
Plants that receive amino acids:
Grow more efficiently
Require less energy to metabolise N
Have fewer pest issues due to lower nitrate accumulation
Often show better mineral balance
Other Sources Of Ammonium
Soil ammonium is not the only source of ammonium to consider but also ammonium produced inside the plant from:
- Photorespiration
- Protein breakdown due to herbicide and pesticide applications
- Senescence
All these sources of ammonium also need to be converted into proteins.
The Risk of Imbalance
When nitrogen isn’t efficiently metabolised:
Nitrate and ammonium build up in the sap
This attracts pests, as many insects can only digest simple nitrogen forms
Photosynthesis gets overwhelmed, especially under stress or poor soil biology leading to lower sugar and carbohydrate levels.
Other minerals become diluted, reducing plant resilience
This is why regenerative nitrogen management is not just about how much nitrogen is applied—but whether the plant can convert it effectively.
Practical Recommendations
Balance nitrogen forms: Ammonium stimulates roots; nitrate drives top growth; amino acids support efficiency.
Ensure co-factors are present: Trace elements like molybdenum, sulphur, magnesium, and nickel are essential.
Monitor sugar levels: Use Brix or sap testing to ensure plants have energy to metabolise nitrogen.
Prioritise amino acids: The most efficient, least stressful nitrogen source for the plant. After amino acids, urea (via foliar), then ammonium and lastly nitrate.
Nitrogen Fertiliser Options
Nitrogen fertilisers play a central role in agriculture but not all nitrogen sources are equal. Some fertilisers are prone to loss, others are energy-intensive for plants to use, and some can even disrupt soil biology or attract pests.
In regenerative systems, the goal isn’t to eliminate nitrogen inputs entirely but to choose forms that are more efficient, plant-friendly, and compatible with soil health. Let’s explore the different nitrogen fertilisers, how they work, and how to use them effectively.
Categories of Nitrogen Fertilisers
Nitrogen fertilisers fall into two main categories:
1. Inorganic Fertilisers
These are synthetic or mineral-based and typically supply nitrogen as:
Urea – 46% N
Highly prone to volatilisation when left on the surface. Needs incorporation or rain to minimise loss from which is it converted into ammonium and nitrate.
In the plant, the urea most be converted into ammonium using the enzyme which contains nickel.
Urea is an excellent foliar options due to it’s cost and ease to disolve, and can be applied at rates of 5-10 kg N/ha, it is recommended to add humic substances to the application.
Ammonium Sulphate (SOA) – 21% N, 24% S
Supplies both ammonium and sulphur.
Ammonium promotes root growth and reproductive development.
Useful where sulphur is needed alongside nitrogen.
Best use: Soil or foliar. Pairs well with urea to support sulphur-containing amino acid production.
MAP (Mono-Ammonium Phosphate) & DAP (Di-Ammonium Phosphate)
Supplies ammonium and phosphorus.
- MAP – 10% N, 22% P
DAP – 18% N, 20% P
Can be used in foliar blends to support early phosphorus needs.
Best use: Starter fertilisers or foliar sprays (at low concentrations).
UAN (Urea Ammonium Nitrate) – 28-32% N
A liquid blend of urea, ammonium, and nitrate.
Delivers a mix of all three nitrogen forms.
Calcium Nitrate – 15.5% N, 18% Ca
- Supplies additional calcium
Can create dilution effects with high water and energy demand due to nitrate.
Best use: Where vegetative growth or tillering is desired; avoid excessive use as it can affect the reproductive growth of the plant.
Potassium Nitrate – 13.8% N, 38.7% K
- Supplies additional potassium
- Similar affects to Calcium Nitrate
2. Organic Fertilisers
These include:
Protein Hydrolysates & Amino Acid Products – 5-10% N
- Contain organic nitrogen in the form of amino acids and peptides.
- Bypass nitrate/ammonium conversion —and directly used in protein synthesis.
- Highly energy efficient and compatible with biological systems.
- Tend to be more expensive then inorganic forms.
- Can be applied as foliars depending on the product
Manures & Compost
Supply a mix of ammonium, nitrate, and organic nitrogen.
Also provide carbon and support soil microbes, in addition to a range of other minerals.
Release nitrogen slowly through microbial decomposition.
N% range depending on the source, can be between 0.5 to 2.5%
Best use: Soil-applied pre-plant or during fallows to build long-term fertility.
Microbial Nitrogen (Rhizophagy Cycle)
Microbes in the rhizosphere are absorbed by plant roots, releasing amino acids and nutrients.
Most energy-efficient and sustainable source of nitrogen.
Requires strong microbial populations and living roots.
Best use: Supported by compost teas, inoculants, cover crops, and low-disturbance soil management.
Nitrogen Budgeting
One of the most common mistakes in nitrogen management, especially in regenerative systems, is cutting fertiliser rates without a solid plan. While reducing nitrogen inputs is a worthwhile goal, doing so blindly can lead to disappointing yields, poor plant health, and financial stress.
That’s where nitrogen budgeting comes in. This process allows you to determine exactly how much nitrogen your crop needs, how much is already in the soil, and how much more you need to apply. In regenerative agriculture, it’s the first step toward reducing inputs without compromising performance.
Why You Need a Nitrogen Budget
Too often, farmers jump straight into regenerative agriculture by cutting nitrogen. While nitrogen overuse is real, the idea that all soils can supply enough nitrogen without inputs is misleading especially in degraded systems.
A nitrogen budget gives you confidence. It removes guesswork, balances soil biology with crop demand, and supports long-term sustainability without short-term losses.
Step 1: Set a Yield Target And Estimate N required
The first step is to define your realistic yield goal and estimate required N.
To determine a yield target, you can:
Use historical farm data or,
Estimate your yield potential based on rainfall and growing conditions (Recommended).
Once we have this number, we estimate the amount of N exported in that product. This can be done using existing removal rates, or calculated from target protein percentage. Using the formula below:
N (kg/t) = P% x 10 x 0.16
Where N (kg/t) is the amount of N as kilograms in each tonne of product, and P% is the protein percentage.
Now we multiply the yield by the removal rate to determine the total amount of N required by the crop per hectare.
Example: For a wheat crop with a 4 t/ha target and a desired grain protein of 13%, you’ll need about 20.8 kg of nitrogen per tonne of yield.
So:
4 t/ha x 20.8 kg N/t = 83.2 kg of nitrogen needed in the grain
This is the physcial amount of N actually in the grain. This N needs to come from somewhere.
But since we typically lose 50% of applied nitrogen, you’ll need to double that to account for losses:
Therefore:
Step 2: Measure How Much Nitrogen Is Already in Your Soil
Use a comprehensive soil test, ideally the Haney Soil Test, to measure:
Nitrate (NO₃⁻)
Ammonium (NH₄⁺)
Organic nitrogen (e.g., amino acids, microbial metabolites)
Example soil test values:
Nitrate: 13 ppm
Ammonium: 18 ppm
Organic N: 20 ppm
Total N = 51 ppm
Now we need to convert this into kg N/ha. To do this use the formula:
Kg N/ha = N in soil (ppm) x Bulk density x depth (cm)/10
Therefore,
51 x 1.4 x 10/10
= 71 kg N/ha
This means our soil contains 71kg of N per hectare to a depth of 10cm.
You can measure deeper into the soil to determine N stored in lower parts of your soil, however, N tends to be greatest in the top 10-15cm. For soils with a deeper top soil, you may wish to sample deeper or have multiple tests.
Step 3: Calculate the Required N
Subtract the nitrogen available from what the crop needs:
This is the amount you’ll need to supply through fertiliser, foliar applications, or biological sources.
Regardless of your system, regenerative or conventional, you need to supply the crop with this additional N otherwise you’ll limit yield.
Step 4: Develop A Nitrogen program
Now we know how much N to supply to our crop, there’s four questions we much ask ourselves in order to develop a nitrogen program, they are the 4 Rs:
- Right time
- Right rate
- Right source
- Right place
In addition to this we also need to know the right price.
Right Time: Match Application to Crop Demand
Plants don’t take up all their nitrogen at once, see image below. Most nitrogen is absorbed during the mid-season growth stages, such as tillering to flowering. Front-loading nitrogen all at once often results in losses, not yield gains.
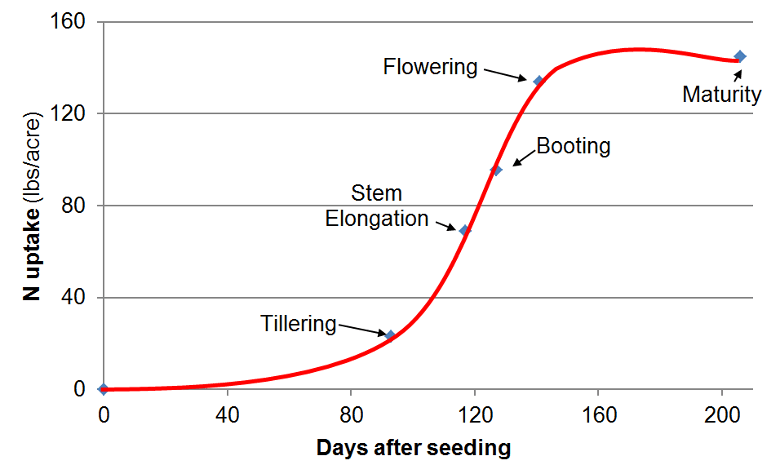
A better strategy:
Split applications: Small amount at planting, more at tillering, and a final top-up if needed.
Foliar applications: Highly efficient, bypass soil losses, and supply nitrogen when and where the plant needs it most.
Right rate, source and place
This well depend on when you’ll apply your N depending on operational limitations, as costs increase with each application.
Source will depend on timing and other minerals required.
Rate will depend on how much to apply at each application and the safe limites of the application.
Placement will depend on equipment, operation and source.
The ideal system would be to apply just enough N to meet the crop’s requirement without over applying, however in the real world there any many limitations to that and nitrogen programs need to be custom designed to suit the goals of each farmer and the context of each farm.
How to Increase Nitrogen Efficiency in Regenerative Farming
In our consulting work, we use the E.S.R framework, it stands for efficiency, substitute and redesign. Meaning we seek to improve the efficiency of the system first, this reduces the risk of losing yield, then substitute for more sustainable sources and then redesign the system to produce it’s own N.
Now that we know how much to apply, and when, let consider how to improve the efficiency of our current fertilisers.
Starter / Side / Top Applications
- Great way of supplying large amounts of N.
- Splitting applications increases N efficiency, as smaller amounts are applied more often, reducing losses and excess N buildup.
- Adding 5% of the application as humic substances. Humic substances contain both negative and positive sites meaning it can hold onto Nitrate and Ammonium while also providing additional carbon to the plant to metabolise the nitrogen. Some estimate that N applications can be reduced by 10-30% as the application of humic substances reduces N loss by 25-40%. To begin, we usually recommend reducing the amount of fertiliser to match the cost of the humic substance.
Foliar Applications
- High efficient way to supply N. Some suggest that foliars are 7-12 times more effective, suggesting that a 5kg N application of foliar N is the equalivate of 50kg N of soil applied N. This really depends on the source and rate of loss, but nevertheless, N supplied as a foliar can be highly efficient.
- Any soluble form of N can be applied with application rates varying depending on the form.
- A typical foliar urea can be applied at 5-10 kg N/ha.
- Always add a carbon source such as humic substances, kelp, molasses to help buffer the nitrogen.
Nitrogen Efficiency Program
A classic way to improve Nitrogen efficiency is by providing all the materials needed to rapidly convert that N into proteins. This can be done either by plants or soil microbes. For any Nitrogen application, apply the following at rates of the nitrogen product:
- 3-5% Humic Substances
- 1-2% Molasses
- 1-2% Kelp
- Sulfur at 10% of N (SOA is a great way to supply N and S)
- 35g/ha Molybdenum
- 10g/ha Nickel (Only if indicated by soil tests that nickel is deficient and foliar urea is being applied.)
Additional Minerals can be applied such as iron and magnesium if required.
Biological Nitrogen Fixation
Microbes can fix nitrogen from the atmosphere and produce ammonium which can then be converted into amino acids and proteins. There’s three groups that can do this:
Symbiotic
These are the microbes that form symbiotic relationships with legumes to fix nitrogen in their roots. These microbes can fix anywhere between 100 to over 300 kg N/ha/yr and supply the majority of a legume’s Nitrogen requirement. These can be utilised in non-legume crops through companion cropping or green manure crops.
Check out our article about Increasing Legume Nitrogen Fixation
Associative
These microbes live in the plant’s rhizophere and consume root exudates in order to fix nitrogen. They include Azospirillum which can fix 10-40 kg N/ha/yr depending on soil health and carbon inputs. These microbes are likely to participate in the rhizophagy cycle.
Asymbiotic
These microbes live freely from plants and can fix around 20kg N/ha/yr. They include Azotobacter and many other species.
How to encourage these microbes:
Maintain root exudates through active photosynthesis
Avoid high synthetic N rates (which suppress fixation)
Provide carbon sources (e.g. humates, molasses)
- Supply minerals required for nitrogen fixation.
- Use inoculants to introduce species and biostimulants to increase activity
Rhizophagy Cycle
Plants can extract amino acids and nutrients directly from microbial biomass. In the rhizophagy cycle, root tips absorb microbes, strip their cell walls, and release nitrogen-rich compounds like amino acids into the plant.
How to support this system:
Maintain living roots year-round
Feed microbes with molasses, seaweed, composts
Inoculate with compost teas or worm extracts
Avoid disruptive inputs (fungicides, high-salt fertilisers)
One study found that up to 33% of a tomato plant’s nitrogen needs could be met by microbial sources under lab conditions.
To read more about the rhizophagy cycle, check out our article James White’s Rhizophagy Cycle

Cover / Catch / Manure Crops
Plants can be planted as part of a nitrogen program to fix nitrogen (either legumes, or plants to feed nitrogen fixing bacteria), or to absorb excess nitrogen currently in the soil to then form plant proteins. Both of these will release that Nitrogen during decomposition where about 50% of that nitrogen will be released each year.
Organic Matter
Organic matter (Soil or otherwise) breaks down to release N. This includes composts, manures, plant residue, animal residue and soil organic matter. Soil organic matter will release at 2-3% each year while the other forms are less stable and might release at about 50% each year.
Measuring Nitrogen In Sap Tests
Soil tests show what’s available in the root zone, but sap tests show what’s actually getting into the plant and how well it’s being metabolised.
Sap testing is especially useful during the growing season to monitor:
Uptake
Excesses or deficiencies
Pest and disease risk
What’s in a Sap:
Total nitrogen
Nitrate (NO₃⁻)
Ammonium (NH₄⁺)
Brix or Sugar %
What to look out for
Compare young and old leaves to determine the translocation of N. If N is being moved from old leaves to young leaves, it suggests that the plant is lacking N, a variation of greater than 10% indicates a deficiency. The total nitrogen should be used to determine nitrogen sufficiency.
This can be used to determine if N needs to be included in a foliar application.
The sap test can also inform Nitrogen metabolism. Ideally there should be 0 ammonium or nitrate in the sap, if there is, then it is likely that protein synthesis isn’t occuring. Sugars, Mg, Mo and S need to be checked to ensure nitrates and ammonium are being converted.
Sap testing will depend on the crop, but it is recommended to sap test during the most important critical points of influence on yields to inform foliar applications.
To read more about differential sap test, check out our article How To Read a Differential Sap Test
Get A FREE Differential Sap Test Analysis!
How Nitrogen Management Affects Pest and Disease Pressure
We often think of nitrogen purely as a growth driver but nitrogen has a profound effect on pest and disease pressure, too. The form, timing, and metabolism of nitrogen can determine whether your crop is healthy and resilient or a magnet for insects and pathogens.
The Link Between Nitrogen and Pest Pressure
Many insects are poor protein digesters. Instead, they rely on plants that are rich in non-protein nitrogen compounds like:
Nitrate (NO₃⁻)
Ammonium (NH₄⁺)
Free amino acids (from poorly metabolised N)
When nitrogen is not properly converted into complete proteins, these free nitrogen compounds accumulate in the sap, making plants:
Easier for pests to digest
More attractive as a food source
More prone to sucking insects, caterpillars, and aphids
Nitrate And Ammonium Accumulation = Pest Pressure
Excess nitrate and ammonium in the plant sap is the #1 contributor to pest susceptibility.
Why? Because nitrate and ammonium are the food source of pests.
If a plant is:
Under stress
Low in trace elements
Lacking energy (low Brix)
then nitrate and ammonium builds up in the sap instead of being used efficiently. This leads to:
High insect pressure
Soft, watery growth
Reduced resistance to pathogens
How to Reduce Pest Pressure Through Smarter Nitrogen Use
1. Improve Nitrogen Metabolism
Support the plant’s internal nitrogen processing by ensuring it has the cofactors it needs:
| Enzyme | Nutrients Required |
|---|---|
| Nitrate reductase | Molybdenum, Iron |
| Nitrite reductase | Sulphur, Iron |
| Glutamate synthase | Magnesium, Manganese |
Also ensure the plant has enough sugars to power the conversion—Brix should be at least 12% for optimal N metabolism.
2. Use Amino Acids Instead of Nitrate
Plants can absorb amino acids directly bypassing the need to convert nitrate. This:
Saves energy
Supports protein synthesis
Limits nitrate accumulation in sap
If amino acids products are too expensive, the next best forms are urea (as foliar), then ammonium.
3. Avoid Overapplication of Nitrogen Fertilisers
Too much nitrate leads to:
Imbalanced plant growth (excess leaf, weak structure)
Dilution of minerals like sulphur, boron, and phosphorus
Higher pest attraction
4. Foliar N Over Soil N
Foliar applications of urea or amino acids are:
More efficient
Less prone to soil losses
Less likely to cause nitrate overload in sap
5. Promote Microbial Nitrogen Supply
Biological nitrogen sources—like microbial metabolites and amino acids—release slowly, support plant metabolism, and reduce pest-susceptible nitrate spikes.
Managing Nitrogen the Regenerative Way
Effective nitrogen management is about more than just applying the right amount of fertiliser, it’s about understanding how nitrogen moves through your soil, how plants use it, and how biology supports or limits that process.
Regenerative nitrogen management focuses on:
Supporting efficient nitrogen metabolism in plants
Reducing reliance on loss-prone synthetic fertilisers
Building stable organic nitrogen pools in the soil
Using biology and monitoring tools to guide decisions
When you align your nitrogen practices with plant physiology and soil biology, you not only save on inputs, you grow healthier crops, build more resilient soils, and reduce pest and disease pressure naturally.
By applying the strategies in this guide, nitrogen budgeting, foliar efficiency, organic sources, biological partnerships, and real-time testing, you can confidently move toward a more sustainable, profitable system that works with nature, not against it.
Get Started In Regenerative Agriculture With Agresol
If you want to implement some of the ideals in this guide on your farm but you’re not sure where to start, reach out for a free consultation and we’ll help you get started!







