In the world of soil science, the Calcium to Magnesium (Ca:Mg) ratio is one of the most debated topics. At Agresol, we often see how a small application of the right nutrient can trigger a massive response, but when it comes to “balancing” these two minerals, the reality is more about physics than a simple recipe.
While we don’t believe in chasing “perfect” numbers for the sake of a spreadsheet, understanding the relationship between these cations is a primary driver of your soil’s physical structure and its ability to “breathe”.
First, watch the video below from our Youtube channel Agresol (and make sure to subscribe!)
The Science of the Clay Colloid
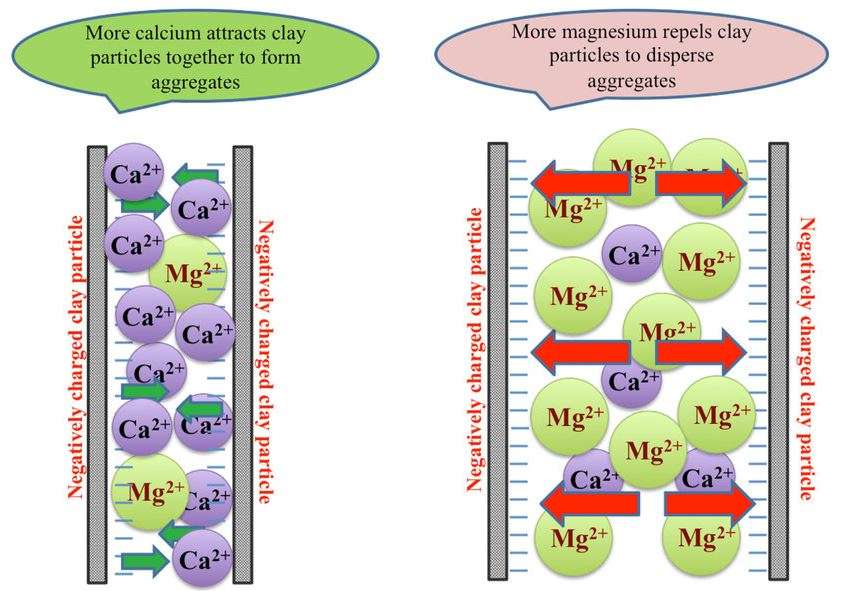
To understand the Ca:Mg ratio, you have to look at your soil under a microscope. Soil contains clay particles, which have negative electrical sites on their outside called colloids. Because opposites attract, positively charged ions, called cations, stick to these colloids like magnets.
The Calcium-Magnesium ratio is simply the balance between the amount of calcium versus magnesium held on these clay active sites.
The Hydration Radius
The reason we care about this ratio isn’t just about nutrition; it’s about physics Although both Calcium and Magnesium have a similar 2+ charge, they behave very differently in the soil:
Calcium (The “Opener”): Calcium is a larger ion. Because it is a “bigger ball,” the force on its outer shell is weaker, meaning it attracts less water. With a smaller hydration radius (42 nm), it can get closer to the clay, pulling particles together tightly in a process called aggregation.
Magnesium (The “Tightener”): Magnesium is a smaller ion with a stronger force that pulls in a lot of water. This creates a larger hydration radius (47 nm). Because the water creates a greater distance between the magnesium and the clay, the “pulling” force is weaker.
The Reality Check: Is There an Ideal Ratio?
Traditional theories suggest specific “ideal” ratios for best soil health:
Sandy Soils: Aim for a ratio of approximately 3:1 (Calcium to Magnesium).
Heavy Clay Soils: Aim for a ratio of approximately 6.4:1.
However, the question remains: does managing for these ratios actually make you money? A large-scale study of 6,300 soil samples found no significant correlation between the Ca:Mg ratio and crop yield. Researchers found high yields in soils with ratios ranging anywhere from 1:1 to 20:1.
The Agresol Management Approach
At Agresol, we recognize that while the ratio is important for soil structure and aggregation, it shouldn’t be your first priority. We use the Ca:Mg ratio as a diagnostic tool, rather than a rigid rulebook, focusing on these key areas:
Addressing Acidity: If your soil is acidic, we use the ratio to decide between applying straight Lime (Calcium) or Dolomite (Calcium and Magnesium).
Managing Sodicity: Excess Sodium causes far worse dispersion than magnesium. In these cases, we apply Gypsum to use calcium to “kick” the sodium off the clay colloid and restore structure.
Prioritizing ROI: Trying to perfectly balance a ratio is often a poor use of funds. You will almost always get a stronger response from foliar applications, seed treatments, or soil primers.
Summary
The Calcium-Magnesium ratio is a window into your soil’s physical health, but it isn’t the final word on your farm’s productivity. We focus on functional results aggregation, infiltration, and plant health, over theoretical “balance”.
If you want to determine if your soil structure is holding back your crop’s potential and find the most cost-effective way to improve your land’s resilience, then sign up for a free 30-minute consultation with Agresol today to review your soil data.







